***********************************
Brakke Consulting’s
Animal Health News & Notes for March 6, 2020
Copyright © Brakke Consulting
Editor: Lynn Fondon DVM MBA
************************************
IN THE NEWS:
Brakke Consulting News
2020 Industry Overview – schedule yours now
Earnings News
Avivagen
Benchmark Holdings
Covetrus
Sanderson Farms
Zomedica
Other News
Addison Biological Laboratory
Afimilk
Akorn Animal Health
Cell BioSciences
Ceva
FarmIQ
Furst-McNess
General Mills
Livestock Improvement Corporation
Mars
Merck (Addison)
Merck (FarmIQ)
Neogen
Nestlé
Pet Plate
VCA
Virbac
Western Nutritional Laboratories
***********************************
SOUTHWEST VETERINARY SYMPOSIUM
Fort Worth, Texas at the Fort Worth Convention Center
September 24-27th, 2020
Early Bird Registration — Now Until July 16th
LEARN MORE. BE MORE.
Registration is NOW OPEN!
Southwest Veterinary Symposium — a partnership of the Arkansas, Louisiana, New Mexico, Oklahoma and Texas Veterinary Medicine Associations – provides continuing education for veterinary professionals. Join us this September in Fort Worth, Texas where you can learn about the latest innovations and products, network with other veterinary professionals, explore design and renovation ideas and choose from more than 400+ hours of quality courses.
************************************
COMPANY EARNINGS RELEASES
- Covetrus, an independent company beginning on February 7, 2019, reported results for the full year 2019. To aid in year-over-year comparability, Covetrus is including certain non-GAAP pro forma financial information that combines the stand-alone Henry Schein Animal Health and Vets First Choice financial information as if the acquisition had taken place on December 31, 2017. Net sales in 2019 were $3,976 million; net loss attributable to Covetrus was $(1,019) million. Net sales in 2018 were $3,778 million; net income for the period was $101 million. (company website)
- Benchmark Holdings reported results for the first quarter of fiscal 2020. Revenues were GBP 25 million ($31.9 million), a decline of 15.5% year-on-year. Net loss for the period was GBP(5.3) million, compared to a net loss of GBP(6.1) million in Q1 2019. (Animal Pharm)
- Avivagen announced its unaudited financial results for the first quarter of 2020. Revenues were C$247,805 (US$185,300), a decrease of C$74,320 over the three-month period ended January 31, 2019. The comprehensive loss from continuing operations was C$(1.3) million. (company press release)
- Zomedica Pharmaceuticals Corp. announced consolidated financial results for the year ended December 31, 2019. Zomedica, which had no recorded revenues, recorded a net loss of $(19.8) million, compared to a loss of $(16.6) million for 2018. The company had cash and cash equivalents of $510,586 as of December 31, 2019, compared to $1,940,265 as of December 31, 2018. (Globe Newswire)
- Sanderson Farms reported results for the first quarter of fiscal 2020. Net sales were $823 million, compared with $743 million for the same period a year ago. The company reported a net loss of $(38.6) million compared with a net loss of $(17.8) million for the 2019 first quarter. (Feedstuffs)
************************************
2020 Brakke Consulting Animal Health Industry Overview
Does your company need a quick, compelling review of what important things happened in the animal health industry in 2019 and what 2020 might look like?
Our annual Industry Overview presented at the VMX and WVC conferences earlier this year can be presented to your company or team and be tailored to fit your specific needs. The key topics this year included the following:
- Industry performance and a special analysis of the public companies in animal health
- Vet practice consolidation, CBD and new companion animal technologies
- Disruptive technologies and the changing landscape of the diagnostics industry
- African Swine fever, plant-based meat and milk products and their impact on protein and animal health product demand
Call one of our consultants (https://brakkeconsulting.com/about-us/our-consultants/), email Amanda McDavid (amcdavid@brakkeconsulting.com), or call her at 336-396-3916 to learn more.
************************************
COMPANY NEWS RELEASES
- Merck Animal Health announced an agreement with Addison Biological Laboratory, Inc., to market the USDA conditionally licensed Moraxella Bovoculi The product is the only commercially available vaccine for the prevention of pinkeye due to Moraxella bovoculi in cattle. (company press release)
- Mars announced that elective declaw surgery is now banned at all 875 VCA Animal Hospitals across the US; only medically necessary declaws will be performed. The policy change follows similar moves at 1,050 Banfield Pet Hospitals and 80 BluePearl practices, also owned by Mars. (Todays Veterinary Business)
- Nestlé announced plans to launch a global pet care accelerator program for early-stage businesses this year. The Unleashed accelerator will identify and support start-ups that “integrate science and technologies into innovative products and services for pets”. Unleashed will leverage Nestlé Purina’s R&D expertise and commercial reach to foster selected start-ups, and provide firms with proof-of-concept funding of up to CHF50,000 ($51,500). (Animal Pharm)
- The FDA announced it has approved Akorn Animal Health‘s generic Pentobarbital Sodium and Phenytoin Sodium Injectable Solution for use in dogs for humane, painless, and rapid euthanasia. (FDA)
- Western Nutritional Laboratories, LLC announced the launch of new Plaque Re-Lease Easy Pinch Tablets for dogs and cats. The Easy Pinch Tablets are made with all-natural cranberry extract and are clinically proven to prevent dental diseases. (company press release)
- Pet Plate, a startup selling freshly cooked pet meals that are flash-frozen before shipping to customers who pay on a subscription basis, announced it has raised $9 million in a Series A round co-led by 301 Inc., the venture capital arm of General Mills, which owns Blue Buffalo pet foods. (Pet Products SmartBrief – Bizjournals)
- The Furst-McNess Company announced the launch of Furst Animal Health brand to better distinguish their health-focused products in the marketplace. Their swine, poultry and dairy products have been in the market for several years. (Hoard’s Dairyman)
- EU The European Medicines Agency’s Committee for Medicinal Products for Veterinary Use (CVMP) announced it has adopted a positive opinion for Ceva‘s Tulaven and Virbac‘s Tulissin – generic injectable tulathromycin products for cattle, pigs and sheep. (Animal Pharm)
- AUSTRALIA Neogen Corporation announced that it has acquired the food safety assets of Australia-based Cell BioSciences, a major supplier of food safety and industrial microbiology products. Cell BioSciences will continue to operate in its non-food safety markets. Financial terms were not disclosed. (company press release)
- NEW ZEALAND FarmIQ, a provider of farm management software, announced that Merck Animal Health has made a minority investment in the company. (scoop.co.nz)
- ISRAEL New Zealand-based cooperative Livestock Improvement Corporation (LIC) announced it has acquired a 50% stake in Israeli dairy farm management business Afimilk. Although some financial details were undisclosed, Afimilk has an enterprise value of $140 million. (Animal Pharm)
***********************************
ANIMAL HEALTH STOCK PRICE TRACKER
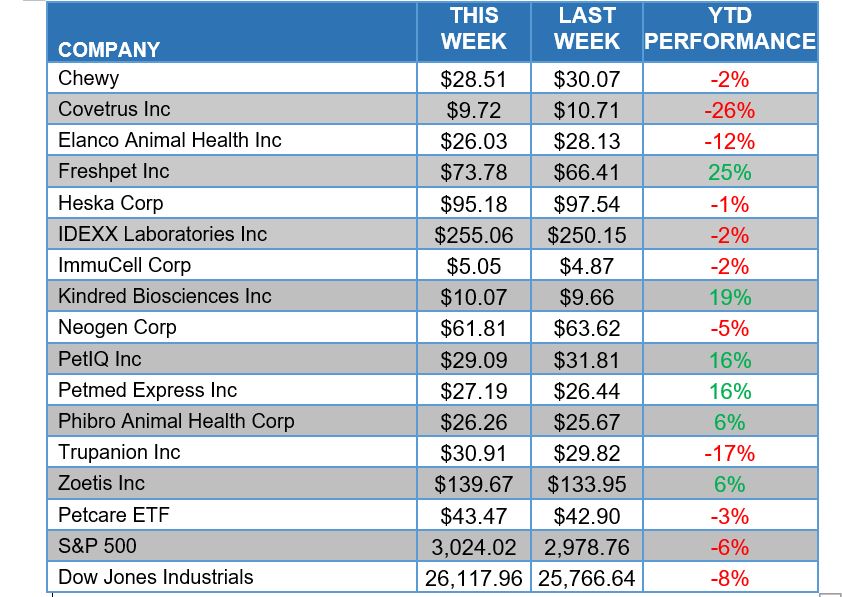
*Thursday market closing prices
***********************************
ANIMAL HEALTH NEWS
- US – COVID-19 In an ongoing evaluation of the impact of coronavirus, the FDA reported that there are 32 animal drug firms that make finished drugs or source active pharmaceutical ingredients in China for the US. Although there are no current shortages, six of those firms – not identified by the FDA – have indicated that they are seeing disruptions in the supply chain that soon could lead to shortages. (Feedstuffs)
- CHINA – ASF RESEARCH The National African Swine Fever Specialty Laboratory of the Harbin Veterinary Research Institute of the Chinese Academy of Agricultural Sciences announced that it has created a live attenuated vaccine against African swine fever (ASF), with good safety and effectiveness for domesticated pigs. After reviewing the paper, the executive director of the Swine Health Information Center stated that there are still “a lot of issues that need to be addressed before this [development] leads to a vaccine,” including the utility of this virus to stimulate immunity against the circulating strain, the possibility of having a “carrier state” of wild virus after vaccination and the lack of being able to differentiate vaccine from wild virus. (Feedstuffs)
- US – HEMP REGULATION The USDA and Drug Enforcement Administration (DEA) reached an agreement that drops the provision in USDA’s Interim Hemp Rule that required hemp growers to test the tetrahydrocannabinol (THC) levels of their crops at a DEA-certified laboratory. Under the new guidance, USDA will delay enforcement of the requirement for labs to be registered by DEA and the requirement that producers use a DEA-registered reverse distributor or law enforcement to dispose of non-compliant plants under certain circumstances. Enforcement will be delayed starting this crop year and until Oct. 31, 2021, or until the final rule is published, whichever comes first. (Feedstuffs)
************************************
BRAKKE CONSULTING VIEWPOINT
Two weeks ago, Bob wrote about COVID-19 and the possible impact on pharmaceutical supply chains. I’d like to expand on this topic and bring up one other concern.
There are reports from Hong Kong that a dog has tested positive for COVID-19 but has shown no clinical symptoms. Currently there is no evidence that dogs or other companion animals play a role in the spread of the disease. Public health experts must quickly and conclusively demonstrate that the current evidence holds true or the impact of panic or evidence-free speculation could create deadly outcomes for many pets.
Regarding supply chains, the FDA reports that 32 animal drug firms make finished drugs or source active pharmaceutical ingredients in China for U.S. sales. Six of those firms have reported seeing supply chain disruptions that will soon lead to product shortages. How soon and for how long will shortages occur? Will current business and regulatory paradigms shift dramatically in response to COVID-19?
I am sure animal health companies will make changes in their supply chains that involve China, but I’m not sure they will rise to the level of true business paradigm shifts in our industry. And that is this week’s question – let us know what you think.
Mike McGinley
***********************************
YOUR VIEW
Last week, we asked what you thought the reduction in the expected 5% growth would be in the animal health industry this year due to the COVID-19 outbreak. Of the respondents, almost half predicted the expected 5% growth would be reduced between 0 and 10% and almost a quarter thought it would be reduced between 21 and 30%. The average reduction was 17%. We’ll get an idea of the impact as first quarter results are announced.
We want to hear from you – share your opinions with us in this one-question poll by clicking below!
This Week’s Question:
**********************************
Trouble viewing this newsletter? You can view it online at www.BrakkeConsulting.com
This electronic newsletter is the sole property of Brakke Consulting.
Any use of the contents herein should be approved by and appropriately attributed to Brakke Consulting.
For more information about Brakke Consulting’s services and syndicated studies, visit our website at www.brakkeconsulting.com.
Brakke Consulting
806 Green Valley Rd
Suite 200
Greensboro, NC 27408
