***********************************
Brakke Consulting’s
Animal Health News & Notes for April 1, 2022
Copyright © Brakke Consulting
Editor: Lynn Fondon DVM MBA
************************************
For All News & Notes Subscribers: At the end of April, we will move to a new platform to host and deliver this newsletter. To keep our subscriber database up to date, we would like you to update your profile, which can be done here. It will take just a minute of your time. We will keep the same well-liked format and will continue sending it out on Friday mornings. Thanks for subscribing – we appreciate it!
Lynn Fondon, Editor
IN THE NEWS:
Brakke Consulting News
2022 Pain Management Products report – pre-order now
Earnings News
Animalcare
Avivagen
Chewy
Kane Biotech
Other News
Apiam Animal Health
Bimeda
Bravo Packing
Elanco
Heska
Norbrook Labs
Percheron Capital
Proteon Pharmaceuticals
Veritas Veterinary Partners
Vetcare Oy
VolitionRx
*********************************************
Brakke Consulting
2022 PAIN MANAGEMENT PRODUCTS FOR DOGS AND CATS
PRE-ORDER BY APRIL 15 FOR DISCOUNT

Pain management is one of the leading topics in veterinary medicine today. Brakke Consulting’s annual report on Pain Management Products for Dogs and Cats includes such valuable information as:
– estimates of US sales and prices of leading products
– new and upcoming pain management products, including ZORBIUM and monoclonal antibody products LIBRELA and SOLENSIA
– discussion of the use of cannabinoids in veterinary pain management
– 2022 survey of 359 small animal veterinarians regarding use of pain management products
NEW THIS YEAR! The report will feature trended, aggregated practice-level data provided by VetSuccess, including chronic vs. acute use of analgesics, comparisons of analgesic revenue per patient by brand, and data on brand-switching at the patient level.
Pain Management Products for Dogs and Cats 2022 can be purchased for $8,750 if ordered by April 15, 2022, and $9,495 thereafter. The report will be completed in late April. Questions about the study can be answered by project manager Lynn Fondon, DVM, MBA at Lfondon@brakkeconsulting.com.
www.BrakkeConsulting.com
************************************
EARNINGS NEWS RELEASES
- Chewy, Inc. announced financial results for the fiscal full year 2021 ended January 30, 2022. Net sales were $8.89 billion, an increase of 24% year over year. Adjusted EBITDA was $78.6 million, a decline of $6.6 million year over year. Net loss was $(73.8) million, including share-based compensation expense of $85.3 million. (company press release)
- Animalcare reported results for the full year 2021. Sales were GBP 74 million ($96.9 million), a year-over-year increase of 5%. Underlying EBITDA improved to GBP 13.5 million ($17.7 million). (IHS Markit Connect)
- Kane Biotech announced its full year 2021 financial results. Total revenue was $1.6 million, a year-over-year increase of 20%. Loss for the year was ($4.8 million), an increase of 26% compared to ($3.8 million) for the year ended December 31, 2020. (company press release)
- Avivagen announced its unaudited financial results for the first quarter of 2022 ended January 31. Net revenues were $271,157, compared to $261,987 in the prior-year quarter. Comprehensive loss was $(1,552,852) compared to a comprehensive loss of $(1,266,300) in the prior-year quarter. (company press release)
*********************************************
COMPANY NEWS RELEASES
- Elanco Animal Health Incorporated announced the commencement of a cash tender offer for up to $250 million aggregate principal amount of its 4.272% Senior Notes due 2023, upon the terms and conditions set forth in the Offer to Purchase dated March 29, 2022. The Offer is scheduled to expire at 12:00 midnight, New York City time, at the end of April 25, 2022. (company press release)
- The FDA announced it has approved Norbrook Laboratories‘ Firox, the first generic firocoxib chewable tablets for use in dogs. (FDA)
- The FDA announced it has approved Finnish company Vetcare Oy‘s Zenalpha (medetomidine and vatinoxan hydrochlorides injection) for use as a sedative and analgesic to help keep dogs sedated and comfortable while undergoing exams or certain medical procedures. (FDA)
- Heska announced an exclusive agreement with VolitionRx to be the sole global distributor of the point-of-care Nu.Q Vet Cancer Screening Test. Under the $28 million license and supply deal, Volition will receive a $10 million upfront payment, as well as up to $18 million based upon achievement milestones. It has also granted Heska non-exclusive rights to sell the test in a kit format for companion animals through its network of central reference laboratories. (IHS Markit Connect)
- The FDA announced that Bravo Packing, Inc., has agreed to stop selling, manufacturing and distributing raw pet food and come into compliance with the Federal Food, Drug, and Cosmetic Act (FD&C Act). Today’s action marks the first consent decree of permanent injunction against an animal food manufacturer for violating public safety standards under Part 507 (Current Good Manufacturing Practice (CGMP) requirements) of the Food Safety Modernization Act (FSMA) Preventive Controls for Animal Food Regulation. (FDA)
- Polish company Proteon Pharmaceuticals announced will enter US market shortly, launching its portfolio of phage-based feed additives that are designed to modulate the microbiome of food-producing animals and enable prophylactic health. (IHS Markit Connect)
- Veritas Veterinary Partners announced it has launched with a national network of over 80 leading specialty and emergency veterinary care providers in the US. Veritas is backed by Percheron Capital. (company press release)
- EU Bimeda announced it has opened a new business unit in Europe to supply products to the Nordic region. The Bimeda Nordic division is headquartered in Odense, Denmark, and will supply primarily large animal products to Denmark, Sweden, Finland, Norway and Iceland. (IHS Markit Connect)
- AUSTRALIA Apiam Animal Health announced it has received a A$700,000 (US$525,000) grant to build the country’s first viral veterinary vaccine manufacturing site. Apiam currently manufactures customized bacterial vaccines via its wholly-owned subsidiary ACE Laboratories, which it acquired in 2019. (IHS Markit Connect)
*********************************************
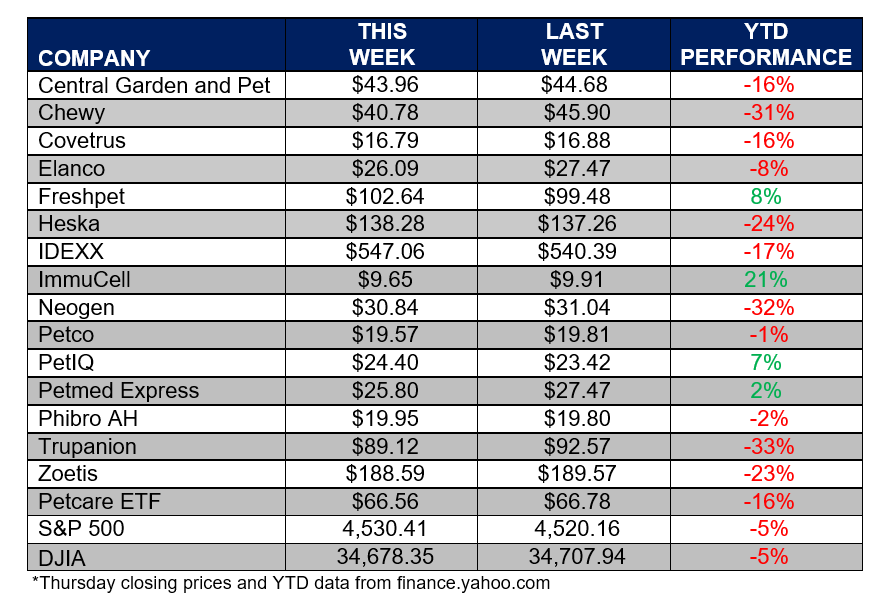
ANIMAL HEALTH STOCK PRICE TRACKER
***********************************
ANIMAL HEALTH NEWS
- US – DRUG APPROVALS The FDA is extending by 60 days the public comment period for the part 15 public hearing on “Scientific Data and Information Related to the Residue of Carcinogenic Concern for the New Animal Drug Carbadox.” The period for public comment is now open until June 10, 2022. To electronically submit comments to the docket, visit regulations.gov and type FDA-2021-N-1326 in the search box. (FDA)
- UK – MASTITIS RESEARCH Researchers at The Roslin Institute have identified a molecule found in milk cells could be used as an accurate way to detect inflammation before clinical signs of mastitis are visible. After studying levels of four types of miRNAs, previously linked to inflammation, they found that three of the four miRNA molecules, known as miR-142, miR-146a and miR-223, could potentially be used for high accuracy, early diagnosis of mastitis before the onset of clinical signs. The study was published in Scientific Reports. (Feedstuffs)
************************************
BRAKKE CONSULTING VIEWPOINT
The lingering effect of the COVID Period and returning to normal has created a lot of discussions recently. As we still face a virus that continues to evolve, we can learn much from the Poultry Industry, which has been battling coronavirus (Infectious Bronchitis) since its isolation in 1931. The first variants were later identified in 1956, and evidence of rapid evolutional ability of the virus has resulted in dozens of known variants that have shown up all over the world. Today it is still considered one of the major disease threats to poultry. But successful vaccine development through the years has helped control the impact of the disease and is always on the forefront of research and monitoring by academia and companies.
In today’s environment with a new focus on ‘One Health’ there could be an excellent opportunity to conduct research and vaccine development between human and veterinary pharmaceutical companies to combat Coronavirus. After all, animal health has a lot of experience to share with our human colleagues.
Paul Casady
***********************************
YOUR VIEW
Last week we asked what has the greatest impact on veterinary practice’s ability to handle increased patient volumes. Most of you said it was due to either a shortage of veterinary technicians (61%) and/or veterinarians (55%); but 44% said it was due to veterinarians wanting to work shorter hours. Here are the complete results:
– 61% shortage of vet technicians
– 55% shortage of veterinarians
– 45% vets wanting to work shorter hours
– 29% continuing COVID-driven protocols slowing patient intake procedures
– 24% supply chain interruptions
– 18% lack of coordination between PMS, inventory and ordering tools
– 11% other
