Brakke Consulting’s
Animal Health News & Notes for September 4, 2020
Copyright © Brakke Consulting
Editor: Lynn Fondon DVM MBA
************************************
IN THE NEWS:
Brakke Consulting News
Mid-Year Overview
Earnings News
Avivagen
Kane Biotech
Patterson Veterinary Supply
Sanderson Farms
Other News
ADM Animal Nutrition
Aurora Pharmaceutical
Bayer
Dechra
DuPont Nutrition & Biosciences
Furst Animal Health
Merck
NovaVive
TriRx Pharmaceutical Services
Vetoquinol
Virbac
Zomedica Pharmaceuticals
************************************
BRAKKE CONSULTING
MID-YEAR ANIMAL HEALTH INDUSTRY OVERVIEW
A big thank you to everyone that attended last week’s 2020 Mid-Year Animal Health Industry Overview. The attendance was greater than we expected, and we appreciate your interest in our views of the growth and resilience of the animal health industry and the hot topics in the companion animal and livestock businesses.
If you would like to have the Mid-Year overview presented to your company, please let us know by emailing Bob Jones at rwjones@brakkeconsulting.com. If you missed it and would like to see a recording, let us know too. Finally, we’d love to hear from you about what topics you’d like us to cover in the next Industry Overview, scheduled for early 2021.
************************************
COMPANY EARNINGS RELEASES
- Patterson Companies, Inc. reported results for its fiscal first quarter ended July 25, 2020. Reported net sales in the Animal Health segment were $812 million, a decline of 0.7% compared to the first quarter of last year. Internal sales for the segment decreased 0.2% from the fiscal 2020 first quarter. (company press release)
- Kane Biotech announced its second quarter 2020 financial results. Total revenues were $267,493, an increase of 21% compared to the prior-year quarter. Loss from operations and finance costs for the second quarter of 2020 was $(505,397), a decrease of 51% compared to the quarter ended June 30, 2019. (company press release)
- Avivagen announced its unaudited financial results for the third quarter of 2020 ended July 31. Revenues were $612,530, an increase of $600,046 over the three-month period ended July 31, 2019. (company press release)
- Sanderson Farms reported results for the fiscal third quarter ended July 31, 2020. Net sales were $956.5 million, compared with $945.2 million for the same period a year ago. Net income was $32.8 million, down from $53.4 million in the 2019 third quarter. (Feedstuffs)
************************************
COMPANY NEWS RELEASES
- The FDA announced the approval of Dechra’s generic carprofen flavored tablets for dogs. (FDA)
- Aurora Pharmaceutical announced the launch of its inaugural companion animal product offering, Revolt (generic selamectin) Topical Parasiticide for Dogs and Cats. (company press release)
- ADM Animal Nutrition announced the launch of Forage First Summer Care, an equine supplement formulated to help working horses combat the effects of heat stress and insects. (Equisearch)
- Furst Animal Health has launched Furst Protect, its first product that specifically focuses to providing nutrients for the immune system. Furst Protect provides pigs, poultry and calves with multiple highly available fatty acids and monoglycerides, along with several natural antioxidants to support and maintain optimum health. (Feedstuffs)
- Zomedica Pharmaceuticals announced it is seeking a reverse split of common shares to continue trading on the NYSE. The proposed consolidation of the company’s outstanding common shares would need shareholder approval. (IHS Markit Connect)
- AUSTRALIA Virbac Australia announced the launch of Cydectin Platinum, the next generation of cattle pour-on drenches. According to Virbac, it is the world’s first dual active moxidectin and levamisole cattle pour-on powered by DMI-Sorb technology, which makes Cydectin Platinum a non-irritant pour-on that ‘stays on’ to enhance penetration and reduce absorption variability of the actives between animals. (PRWire.com.au)
- EUROPE Vetoquinol announced the completion of the acquisition from Bayer of Profender and Drontal product families for the European Economic Area and the UK. (Business wire)
- FRANCE TriRx Pharmaceutical Services, a global contract development and manufacturing organization (CDMO), announced that it has finalized an agreement with MSD Sante Animale, a division of Merck, to acquire the operations and ownership of their manufacturing site located in Segré, France. Financial terms were not disclosed. (PRnewswire)
- NEW ZEALAND NovaVive, announced that Amplimune, its immunotherapy for calf scours, has received approval for sale in New Zealand by Agricultural Compounds and Veterinary Medicines (ACVM). (company press release)
- CHINA DuPont Nutrition & Biosciences announced the opening of a new animal nutrition manufacturing facility in Wuxi, China. DuPont Animal Nutrition provides nutritional health solutions with a comprehensive portfolio of feed enzymes, betaine, phytogenics and probiotics. (Feedstuffs)
***********************************
ANIMAL HEALTH STOCK PRICE TRACKER
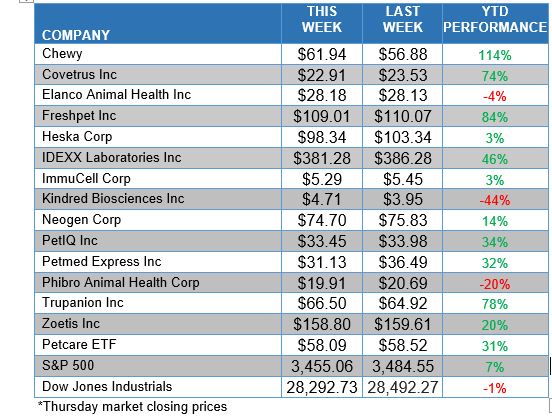
*Thursday market closing prices
***********************************
ANIMAL HEALTH NEWS
- US – FELINE VACCINATION The American Animal Hospital Association (AAHA) and the American Association of Feline Practitioners (AAFP) announced the release of the 2020 AAHA/AAFP Feline Vaccination Guidelines, which provides updated recommendations and the most current information for feline vaccinations. To read the guidelines, click here. (association press release)
- US – VETERINARY UNION The veterinary industry’s first-ever private-sector union contract has been ratified at a Vancouver, Washington clinic. The agreement, which aims to retain qualified employees at Columbia River Veterinary Specialists (CRVS) by ensuring its hospital workers are fairly compensated and protected, was approved by a margin of 53 to 1. CRVS employees had been in negotiations with the hospital’s management team since voting to join the International Longshore and Warehouse Union (ILWU) Local 5 in February 2019. (Veterinary Practice News)
************************************
BRAKKE CONSULTING VIEWPOINT
It is hard to see opportunities when you’re looking through a pandemic, but there is one right now for U.S. pet food manufacturers – the booming pet food market in China.
The regulation on exporting pet food products to China has been streamlined since the Phase I Economic and Trade Agreement between the U.S. and China was signed in January this year. Two significant changes from the past: first, China has lifted its ban on U.S. pet food containing ruminant ingredients; and second, the process to get manufacturing facilities on the list of approved exporters with the General Administration of Customs of China (GACC) is shortened from 2-3 years to 1 month. Dependent on the types of pet food product, FDA or APHIS will submit facility update to GACC.
Facilities may need to be inspected by USDA APHIS if exporting regular pet food, including treats and chews, and there are certain exemptions to inspections. These changes provide a great opportunity for U.S. pet food product manufacturers that are interested in the fast growing petcare market in China, where pet owners are looking for high quality pet foods. If you have questions or if we can help in any way, please contact us.
Jishu Shi
***********************************
YOUR VIEW
Last week, we asked you about how well you thought your company was adapting to doing business and being successful during the pandemic. Just over half of the respondents gave their company an “A” – excellent. About a third gave their company a “B” and the balance gave their company a “C”. No poor or failing grades. As Randy Freides mentioned in our Mid-Year Industry Overview last week, being able to adapt to changes in business is one of the definitions of resilience.
This week
This week let’s pick up on a topic from two items in this week’s newsletter – the use of generics drugs in veterinary medicine.
