***********************************
Brakke Consulting’s
Animal Health News & Notes for July 31, 2020
Copyright © Brakke Consulting
Editor: Lynn Fondon DVM MBA
************************************
IN THE NEWS:
Brakke Consulting News
COVID-19: Impact on Pet Care report – wave 3 coming soon
Earnings News
Alivira Animal Health
Elanco
Nestle
Vetoquinol
Other News
ADM Animal Nutrition
AKL Research and Development
Boehringer Ingelheim
Canapp Sports Medicine
Cargill
Ceva Sante Animale
Cubex
Fresenius Kabi
Global Stem cell Technology
Midmark
Nestlé Purina PetCare
Plug & Play
Protenga
Roslin Technologies
Trice Medical
Virbac
************************************
BRAKKE CONSULTING
COVID-19: IMPACT ON PET CARE – Wave 3 available in mid-August
HOW HAVE PET OWNERS CHANGED THEIR PURCHASING?
The pandemic that struck the US and the rest of the world in early 2020 is an event that has altered personal lives and business operations unlike any other event. What lasting impacts will the pandemic have on pet owners regarding the way they purchase pet medications and veterinary services?
“COVID-19: Impact on Pet Care” is a new study by Brakke Consulting that will help answer this question. The study is based on online surveys of a nationally representative sample of 1,000 pet owners, conducted every few weeks beginning in May. The study not only documents the financial impact of the COVID-19 pandemic on pet owners, it also shows the change in how pet owners were able to receive care for their pets at veterinary clinics. Furthermore, it shows how pet owner purchasing behaviors for pet medications and pet food changed during the stay-at-home orders.
The study delivers 3 reports, roughly 4 – 6 weeks apart, beginning in May; the final report in the series will be available in mid-August. The cost of the study is $17,500. For more information, contact John Volk at john@volkonline.com .
************************************
COMPANY EARNINGS RELEASES
- Elanco Animal Health reported financial results for the second quarter of 2020. Total revenue was $586 million, a decrease of 25% compared with the second quarter of 2019 (-23% without the impact of foreign exchange rates). Net loss was $(53.2) million compared with net income of $35.9 million for the same period in 2019. (company press release)
- Vetoquinol SA reported results for the first half of 2020. Total sales were EUR 196 million ($220 million), an increase of 7.5% at constant exchange rates. Net income was EUR 15.1 million ($17 million), an increase of 16% compared to the prior-year period. (company website)
- Nestle SA reported results for the first half of 2020. PetCare sales were CHF 6.98 billion ($7.36 billion), representing real internal growth of 11%. (company website)
- Alivira Animal Health reported results for the first quarter of fiscal year 2021 ended June 30, 2020. Revenues were $41 million, an increase of 12% compared to the prior-year quarter. Net profit was $3.2 million compared to a net profit of $2.0 million in the prior-year period. (company press release)
************************************
COMPANY NEWS RELEASES
- Boehringer Ingelheim announced it has acquired Belgium-based Global Stem cell Technology (GST), a company dedicated to the research, development and production of stem cell therapies used to treat orthopedic and metabolic diseases in animals. Boehringer Ingelheim already entered into a partnership with GST in 2018, and in 2019, the companies launched Arti-Cell Forte in Europe. Financial terms were not disclosed. (Business Wire)
- The FDA has approved Ceva Sante Animale‘s Cardalis (spironolactone and benazepril hydrochloride chewable tablets) for use with other medications (e.g., diuretics) for the management of the clinical signs of mild, moderate or severe congestive heart failure in dogs due to a form of heart disease in dogs known as atrioventricular valvular insufficiency (AVVI), which can lead to congestive heart failure. Cardalis is available in three sizes of flavored tablets. (FDA)
- Nestlé Purina PetCare announced the launch of Purina Pro Plan Veterinary Diets Urinary Health, a veterinary-exclusive treat formulated to assist with feline urinary health. (Vet Advantage)
- The FDA announced a recall of two lots of Fresenius Kabi’s Dexmedetomidine HCl in 0.9% Sodium Chloride Injection, 200 mcg/50 mL vial due to the possible presence of trace amounts of lidocaine in these two lots based on FDA investigation. To date, no adverse drug experience reports have been received for either of the lots being recalled by Fresenius Kabi. (FDA)
- Archer Daniels Midland (ADM) Animal Nutrition announced the launch of NutriPass L encapsulated lysine product for beef and dairy cows in the US. (IHS Markit Connect)
- Midmark introduced the Midmark Multiparameter Monitor, featuring Cardell blood pressure (BP) technology, veterinary-specific algorithms and innovative staff-friendly capabilities. Each monitor is equipped with the accessories necessary to allow for full monitoring capability, including BP cuffs, probes, sensors and connecting cables appropriate for the parameters chosen. (company press release)
- Trice Medical announced it has partnered with Canapp Sports Medicine LLC to launch the world’s first veterinary portable tablet combining both a disposable needle arthroscope and high-quality ultrasound into one system. The mi-eye needle arthroscope (with a 1.8mm camera) and mi-ultra ultrasound together are used on one single, portable HD tablet. (PRnewswire)
- Cubex LLC announced the release of CubexPMP, software designed to automate the recording and submitting of prescriptions under state-run Prescription Monitoring Programs. Through integration with veterinary practice management software, all transactional data is electronically captured and sent to CubexPMP, and complete, accurate records are submitted automatically in the timeframe required by the state. (Todays Veterinary Business)
- Silicon Valley-based tech company, Plug & Play, announced that Cargill will serve as a founding partner for its newest location in Topeka, Kansas focused on animal health and agriculture technology. Through this partnership, Plug and Play and Cargill will work with startups that are creating new technologies and products to spur innovation in the animal health and ag tech industry. (company press release)
- UK Virbac announced the UK launch of Tramvetol, licensed tramadol flavored tablets for the treatment of acute and chronic pain in dogs. (Vetsurgeon.org)
- UK AKL Research and Development announced it has signed an agreement with one of the world’s top five animal health companies to fund the development of its next-stage clinical trial of an investigational drug called APPA in the treatment of canine osteoarthritis (OA). APPA is an oral, patented, fixed-dose combination of two synthetic secondary metabolites of plant origin, apocynin and paenol. (Vetsurgeon.org)
- SINGAPORE Roslin Technologies announced an investment in Singapore-based Protenga, an innovative business that farms black soldier fly insects to produce animal feed and fertilizer. As part of Roslin’s program, a bespoke genetic nucleus facility is being built near its headquarters in Edinburgh, Scotland, that will focus on applying enhanced breeding technologies to develop improved insect lines with Protenga for the global market. (Feedstuffs)
***********************************
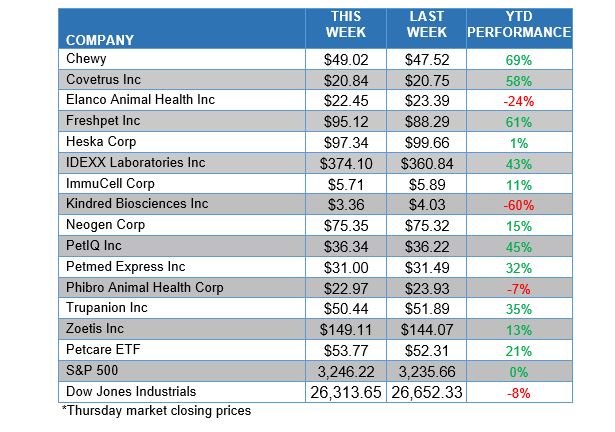
ANIMAL HEALTH STOCK PRICE TRACKER
 ***********************************
***********************************
ANIMAL HEALTH NEWS
- US – BRD TESTING Purdue University announced it has received a $1 million USDA grant to develop rapid sensor technology for bovine respiratory disease (BRD). A team of researchers at the university are working to reduce the diagnosis time of BRD to about 30 minutes. Standard testing can take days to generate results. (IHS Markit Connect)
- US – DRUG ADVERSE REACTION REPORTING The FDA has issued the final rule to require animal drug sponsors to submit to the agency certain adverse drug experience and product manufacturing defect reports on Form FDA 1932 in electronic format. (FDA)
- US – PRACTICE GUIDELINES The AVMA has added a new tool to its COVID-19 resource page to help practices safely expand services during the pandemic. The risk management guide covers topics like employee health, safety and compliance; premises liability; client safety; and property loss prevention. It also includes other AVMA tools, such as an employee screening template and a “case management decision tree,” which helps veterinarians decide when it’s safe to see patients and when postponing or referring is safer. (Vet Advantage)
************************************
BRAKKE CONSULTING VIEWPOINT
This is usually the time of year when our industry is reconnecting with one another at one of several conferences – the AVMA Convention, the KC Animal Health Corridor Homecoming and Investment Forum, or one of the Fetch summer conferences. Thanks to COVID-19, all that has changed, and these events have moved to virtual formats later in August (the KC Corridor events have been rebranded this year as the Digital Animal Health Summit). What kind of opportunities for networking will be available at these virtual conferences, and will attendees get the kind of person-to-person interactions they are looking for? We’ll find out soon.
Several of our consultants will be participating in the Digital Animal Health Summit on August 31 – September 2, so we will be part of this experiment. We hope to connect with many of you then.
Lynn Fondon
***********************************
YOUR VIEW
Last week we asked “What has been the effect of the last six months of the COVID-19 situation on animal health innovation and product development?” Two-thirds of you feel that the pace of innovation has slowed, although 18% feel that it has accelerated. Our newsletter has mentioned a few companies over the past couple of months that have jumped into helping find solutions for the coronavirus crisis, from repurposing manufacturing facilities to shifting product development to a possible COVID-19 vaccine or treatment. However, for most companies the focus currently may be on maintaining sales volume or allocating resources to make workplaces safer, rather than developing new products.
This week:
Let’s spend a minute thinking about leisure instead of work. Summer is almost over, and what a crazy time it’s been. Did you take a summer vacation this year?
**********************************
Trouble viewing this newsletter? You can view it online at www.BrakkeConsulting.com
This electronic newsletter is the sole property of Brakke Consulting.
Any use of the contents herein should be approved by and appropriately attributed to Brakke Consulting.
For more information about Brakke Consulting’s services and syndicated studies, visit our website at www.brakkeconsulting.com.
Brakke Consulting
806 Green Valley Rd
Suite 200
Greensboro, NC 27408
