***********************************
Brakke Consulting’s
Animal Health News & Notes for August 21, 2020
Copyright © Brakke Consulting
Editor: Lynn Fondon DVM MBA
************************************
IN THE NEWS:
Brakke Consulting News
COVID-19: Impact on Pet Care – final report now available
Mid-year Overview Webinar
Earnings News
Better Choice Company
Other News
BabelBark
Doggie Dailies
Fidelis Pharmaceuticals
Impossible Foods
LexaGene
LaunchWorks
Manna Pro Products
MWI Animal Health
Petmate
Pet Qwerks
Viticus Group
************************************
BRAKKE CONSULTING
COVID-19: IMPACT ON PET CARE – final report now available
HOW HAVE PET OWNERS CHANGED THEIR PURCHASING?
The pandemic that struck the US and the rest of the world in early 2020 is an event that has altered personal lives and business operations unlike any other event. What lasting impacts will the pandemic have on pet owners regarding the way they purchase pet medications and veterinary services?
“COVID-19: Impact on Pet Care” is a new study by Brakke Consulting that helps answer this question. The study is based on online surveys of a nationally representative sample of 1,000 pet owners, conducted roughly every six weeks beginning in early May. The study not only documents the financial impact of the COVID-19 pandemic on pet owners, it also shows the change in how pet owners were able to receive care for their pets at veterinary clinics. Furthermore, it shows how pet owner purchasing behaviors for pet medications and pet food changed during the stay-at-home orders.
The study delivered 3 reports, roughly 6 weeks apart, beginning in May; the final report in the series is now available. The cost of the study is $17,500. For more information, contact John Volk at john@volkonline.com .
************************************
COMPANY EARNINGS RELEASES
- Better Choice Company, Inc. reported its financial results for the second quarter ended June 30, 2020. Net revenue was $9.9 million, a 140% increase over the comparable period in 2Q19. The majority of this increase is attributable to the Halo acquisition. Net loss was $(18.4) million compared to a net loss of ($161.5) million in the prior-year period. (Globe newswire)
************************************
BRAKKE CONSULTING
The 2020 Midyear Industry Overview Webinar
Date: August 28, 2020
Time: 11:00 am – 12:30 pm EDT
The Midyear Overview will check in on top animal health company performance and key operational highlights for the first half of the year, especially in light of the lockdowns due to COVID-19. We will highlight public animal health company investment performance and explore how resilient animal health companies have been given the economic pressures of the pandemic and will describe what the winners have done to be successful. The presentation will include new, unpublished data tracking the impact of COVID-19 on pet care as owners have weathered upheaval in their daily lives, plus some fascinating data on the performance of veterinary practices, including their use of telemedicine and home delivery. Finally, we look at how the livestock business has been directly and indirectly impacted by the lockdowns and what this means for future growth.
This timely webinar review will be useful as animal health and nutrition leaders prepare for the second half of the year and start on their budgets for next year. Brakke Consulting believes that events like the COVID-19 pandemic tend to accelerate trends that were already happening. But the response to the pandemic has also resulted in actions not commonly seen in our animal health and nutrition industry. Will these changes last into next year or beyond? In 90 minutes, the Brakke Consulting team will present the first half highlights and its outlook.
Date: August 28, 2020
Time: 11:00 am EDT
Cost: $99
Signup for the webinar by clicking here.
************************************
COMPANY NEWS RELEASES
- Manna Pro Products announced it has acquired Doggie Dailies, an online pet brand that offers premium nutritional supplements and wellness products for dogs. Financial terms were not disclosed. (Pets International)
- Fidelis Pharmaceuticals announced the availability of new Ethiqa XR (buprenorphine extended-release injectable suspension) for the control of post-procedural pain in mice and rats. Ethiqa XR provides up to 72 hours of pain relief with just one injection. Laboratory veterinarians and researchers can order Ethiqa XR on a research DEA license (and any applicable state license or registration) directly from distributors, including MWI Animal Health. (company press release)
- Petmate announced it has acquired Pet Qwerks , a company that designs and develops quality dog chews and interactive toys for dogs and cats. Financial terms were not disclosed. (Pet Business)
- LexaGene announced it has partnered with contract manufacturer LaunchWorks to produce the consumables needed to operate its MiQLab molecular diagnostics system. MiQLab is LexaGene’s first commercial product and is scheduled to be launched before the end of September 2020. The fully automated rapid pathogen detection technology is for use in animal health, human health and food safety testing. (IHS Markit Connect)
- The International Association of Animal Hospice and Palliative Care (IAAHPC) announced a partnership with BabelBark, Inc., offering asynchronous two-way pet journaling to assist veterinary care providers and pet parents when hospice and palliative care is needed. (company press release)
- Viticus Group announced that the next WVC Annual Conference has been rescheduled from February to September 2021. Viticus Group is also adding a virtual component to the 2021 conference, making it their first hybrid event. (Vet Advantage)
- Impossible Foods announced it has secured $200 million in its latest funding round. The food tech startup has raised about $1.5 billion since its founding in 2011. (Feedstuffs)
***********************************
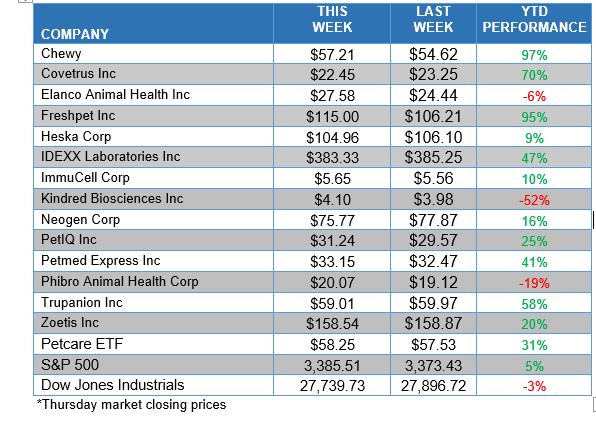
ANIMAL HEALTH STOCK PRICE TRACKER
***********************************
ANIMAL HEALTH NEWS
- US – SARS-CoV-2 The USDA’s National Veterinary Services Laboratories (NVSL) announced the first confirmed cases of SARS-CoV-2 (the virus that causes COVID-19 in humans) in mink at two farms in Utah. These are the first confirmed cases of SARS-CoV-2 in mink in the US, and the affected farms also reported positive cases of COVID-19 in people who had contact with the mink. (Feedstuffs)
************************************
2020 DIGITAL ANIMAL HEALTH SUMMIT
AUGUST 31 – SEPTEMBER 2, 2020
Location: Online
Registration closes Monday, August 24

The Corridor’s August event goes on(line)! Registration is now open.
Register today ! The digital Animal Health Summit features 36 hours of 1:1 business partnering, keynote speaker David Hughes, 7+ on demand webinars, emerging company presentations, LIVE stream programming with industry leaders, LIVE networking and additional programming that supports the Spirit of Service – programming with impact. Click here to learn more.
************************************
BRAKKE CONSULTING VIEWPOINT
In line with my colleague’s comments last week, it is quite astonishing how the animal health industry keeps moving positively despite the difficult times caused by the pandemic. But for those of us in the industry, it is not really that surprising. I wonder if we can take some of this positive performance and really challenge ourselves to play a stronger role in public health. Lots of good people out there have good ideas. And there will be another pandemic someday; maybe animal health can limit or neutralize the threat!
On a sad and personal note, we recently lost an animal health veteran and good guy, Steve Vandeberg, to COVID-19. Steve had a long career in animal health between Merck (MSDAgVet) and later Merial. He was a consummate professional and recognized marketing expert in large animal endectocides. We will all miss him.
Take care everybody,
Paul Casady
***********************************
YOUR VIEW
Last week we asked, “How easy is it for animal health companies to market, sell and support both products and services?” A whopping 82% of respondents thought that it wasn’t that easy since products and services require very different strategies and tactics to be successful. An equal minority of 9% each thought it was easy to do both and thought it is not possible to do – you needed to be good at one or the other. We’d agree – it is not that easy.
This week
This week, let’s focus on vaccines and how they are used to prevent highly infectious diseases. In animals, Foot and Mouth Disease (FMD) is a highly infectious, sometimes fatal disease of cloven-hoofed animals. To prevent its spread in FMD-endemic countries, vaccines are used and often mandated by the government. The rapid development of human vaccines for SARS-CoV-2 (the virus that causes COVID-19) is in the news daily and companies hope to have them available in months. State governments will soon decide on how they will be used or mandated, especially for entrance into schools, where vaccines to certain diseases are already required for entrance.
This week’s question,
Once an FDA-approved vaccine for SARS-CoV-2 is available, should schools require it for attendance?
